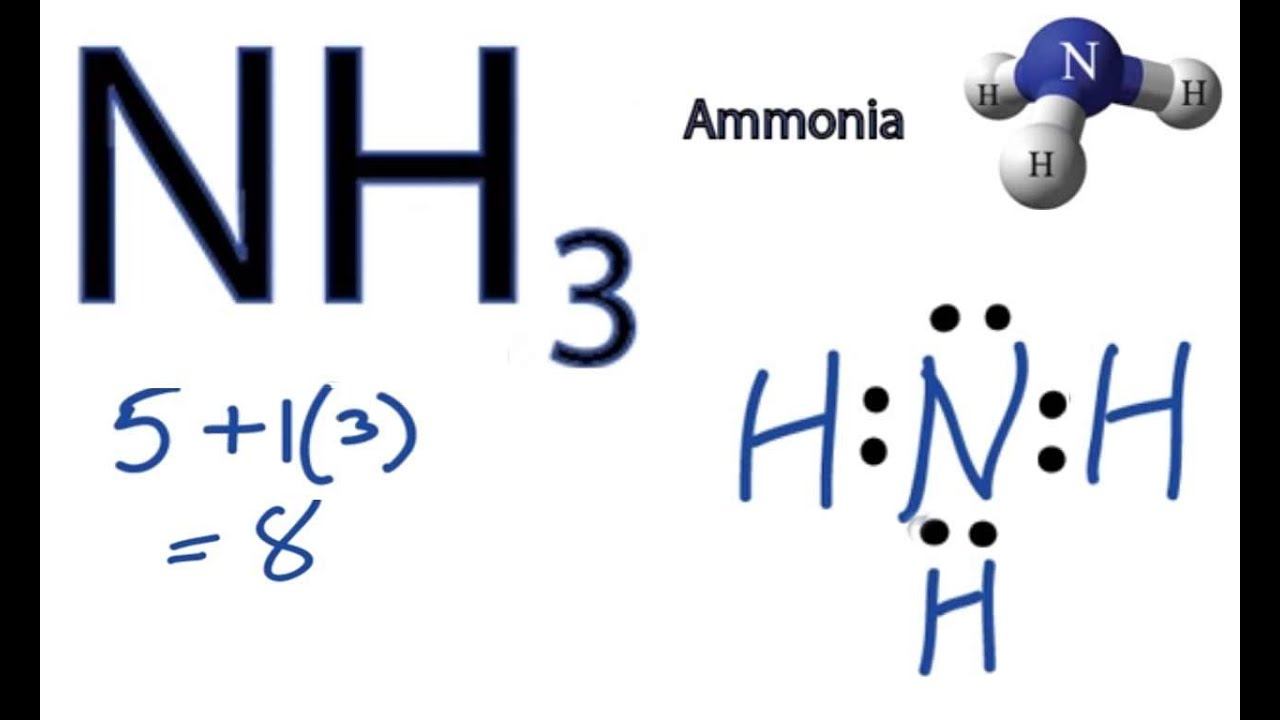
Lewis Dot Diagram For Nh3 exatin.info
1: Structure and Bonding 1.3: Lewis Structures Expand/collapse global location 1.3: Lewis Structures Page ID Using Lewis Dot Symbols to Describe Covalent Bonding

NH3 Molecular Geometry Science Education and Tutorials
= 8 valence Ammonia or NH3 has a total of 8 valence electrons. NH3 Lewis Structure The Lewis structure of a molecule helps understand the electron geometry, molecular geometry, polarity, and other such properties with ease. It is a pictorial representation of the arrangement of valence electrons around the individual atoms in the molecule.
Lewis Dot Diagram For Nh3 exatin.info
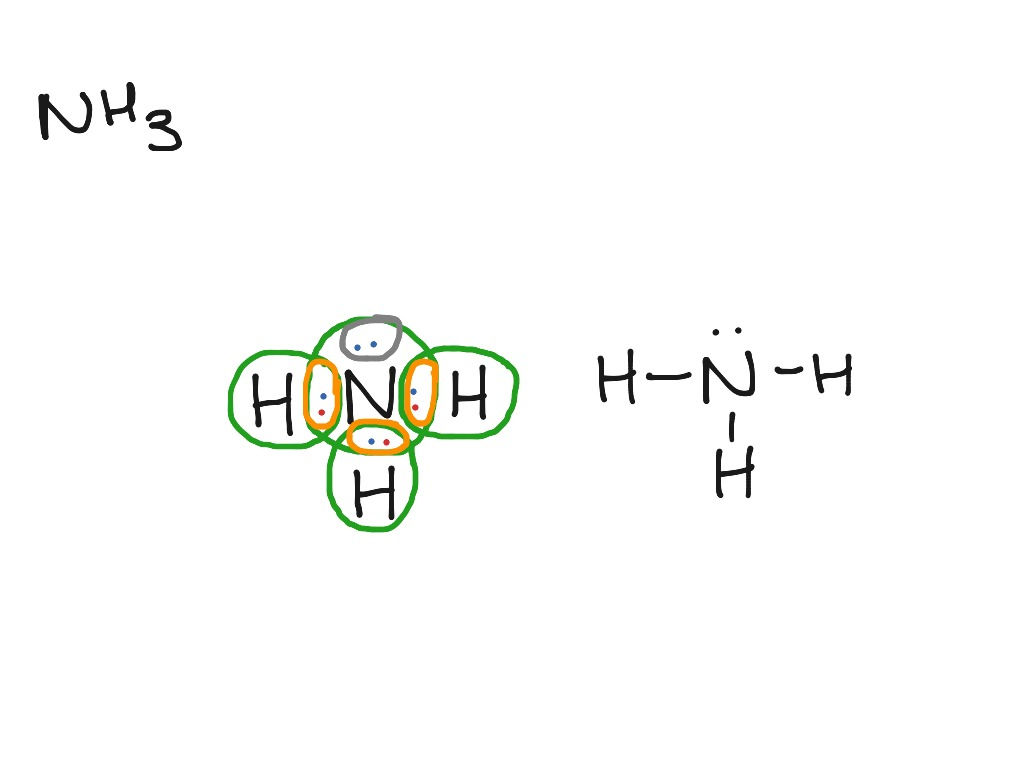
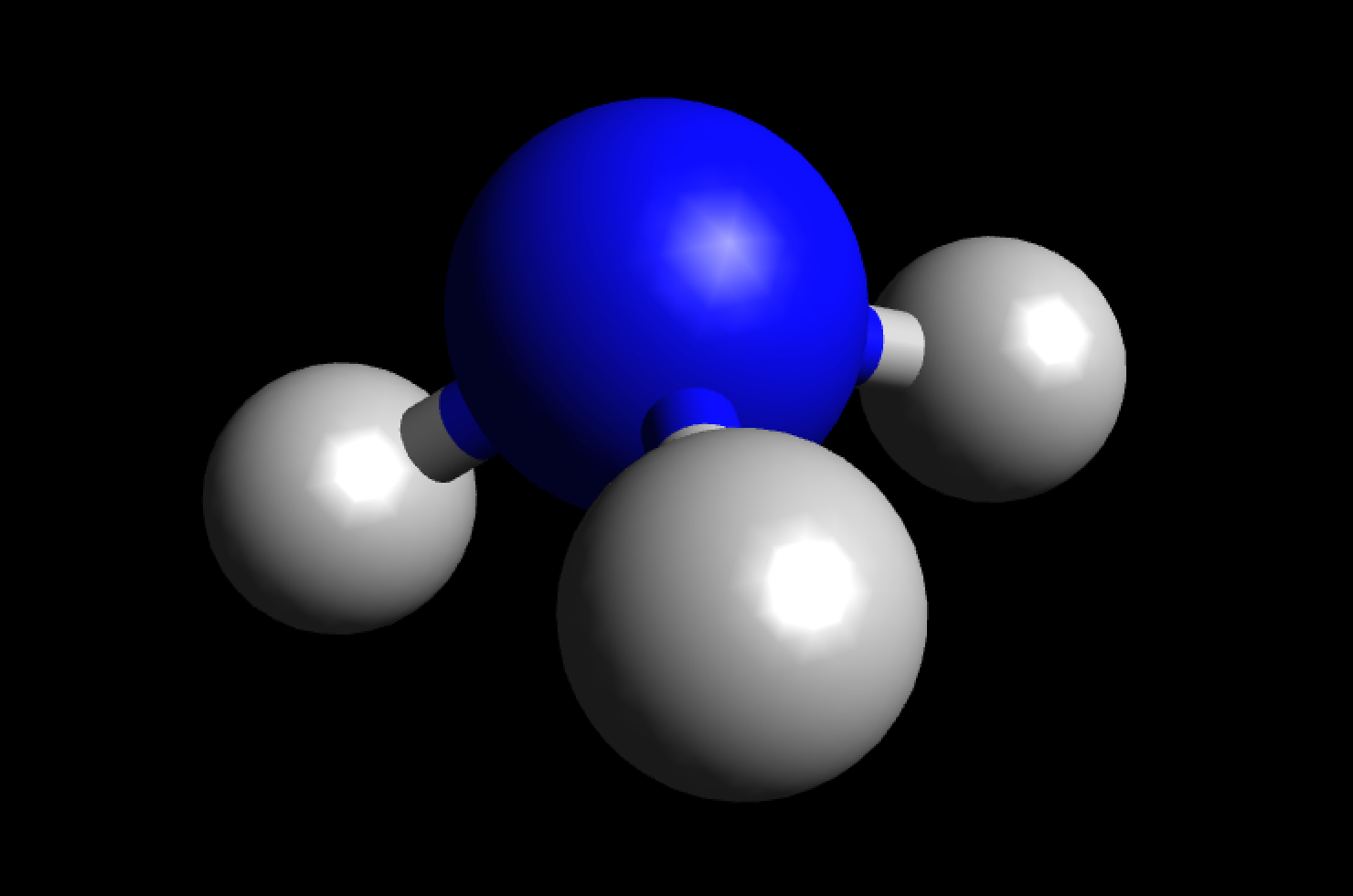
Lewis structure of NH3 (ammonia) contains three single bonds between the Nitrogen (N) atom and each Hydrogen (H) atom. The Nitrogen atom (N) is at the center and it is surrounded by 3 Hydrogen atoms (H). The Nitrogen atom has one lone pair. Let's draw and understand this lewis dot structure step by step.

17 Beautiful Nh3 Lewis Structure 3d Model Viela Mockup
Steps of drawing NH3 lewis structure Step 1: Find the total valence electrons in NH3 molecule. In order to find the total valence electrons in NH3 molecule, first of all you should know the valence electrons present in nitrogen atom as well as hydrogen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)

Lewis Dot Diagram Of Nh3
NH3 Lewis structure: Steps involved in the NH3 Lewis structure: Step 1: Valance Electron Determination: Step 2: Central Metal Atom: Step 3: Connect the Atoms with a lone pair: Step 4: Distribute Lone Pairs: Step 5: Complete the Lewis Structure: Step 6: Formal Charge: Detailed applications: Synthesis/Production:
3d nh3
Lewis Structure Finder. This widget gets the Lewis structure of chemical compounds. Get the free "Lewis Structure Finder" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha.
Lewis Dot Diagram For Nh3 exatin.info
To begin drawing the NH3 Lewis structure, start by counting the total number of valence electrons. Valence electrons are the outermost electrons of an atom and are involved in bonding. For NH3, nitrogen (N) is in Group 5A (Group 15), so it has five valence electrons.

Lewis Structure of NH3 YouTube
Ammonia (NH 3) is a commonly tested Lewis structure due to it's widespread use in agriculture as a fertilizer. It also is a good example of a molecule with a trigonal prymidal molecular geometry. There are 8 valence electrons available for the Lewis structure for NH 3. Video: Drawing the Lewis Structure for NH3 It is helpful if you:

Is NH3 Polar or Nonpolar?Is Ammonia a Polar or Nonpolar Molecule?
A video explanation of how to draw the Lewis Dot Structure for Ammonia, along with information about the compound including Formal Charges, Polarity, Hybrid.

Estrutura De Lewis Nh3 AskSchool
In the NH3 lewis dot structure, there are Four atoms present, one N and three H atoms. The valence electrons for N are 5 and for three H atoms are 3. So, the total valence electrons are 5+3 = 8. Step 2 - According to the octet rule, the electrons that will be needed for the NH3 lewis dot structure will be 8 + (3*2) = 14.
MakeTheBrainHappy Is NH3 Polar or Nonpolar?
There is really only one way to draw the Lewis structure for Ammonia (NH3). For resonance structures there must be a double or triple bond present, which is.
Lewis Dot Diagram For Nh3 exatin.info
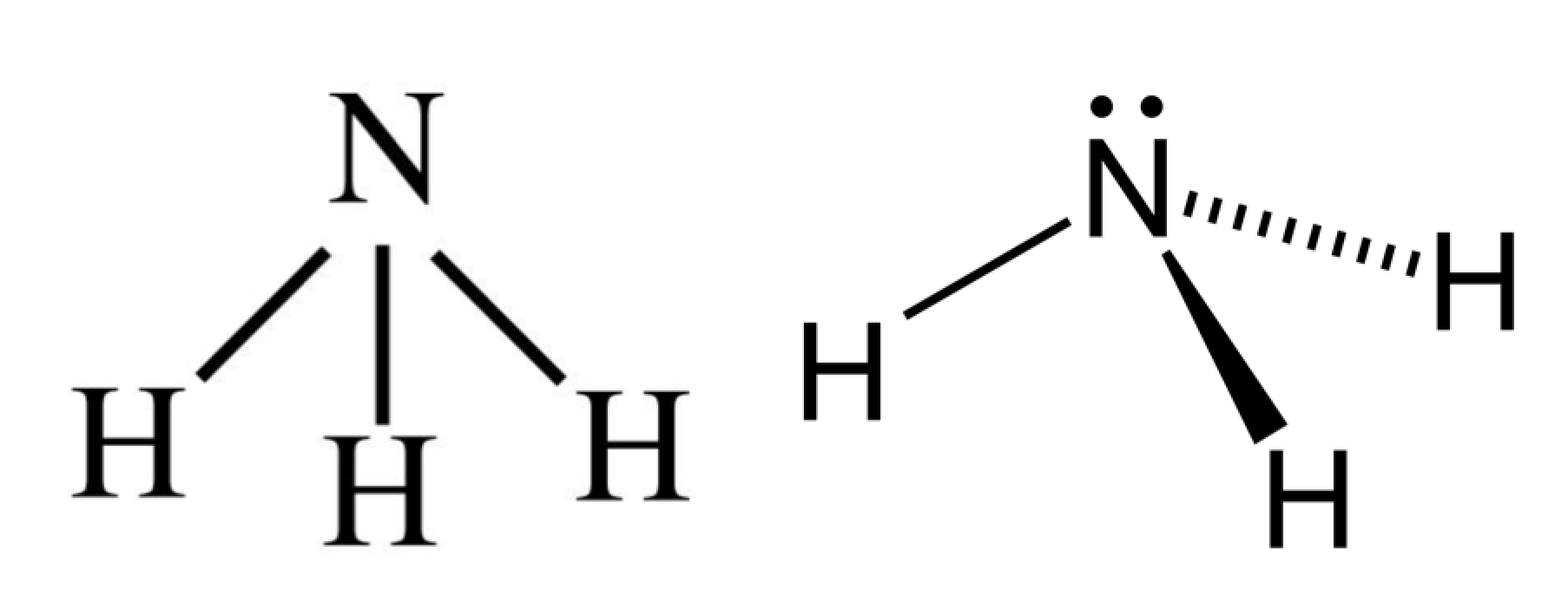
The Lewis structure of ammonia, NH_3, would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons.

How to draw NH3 Lewis Structure? Science Education and Tutorials
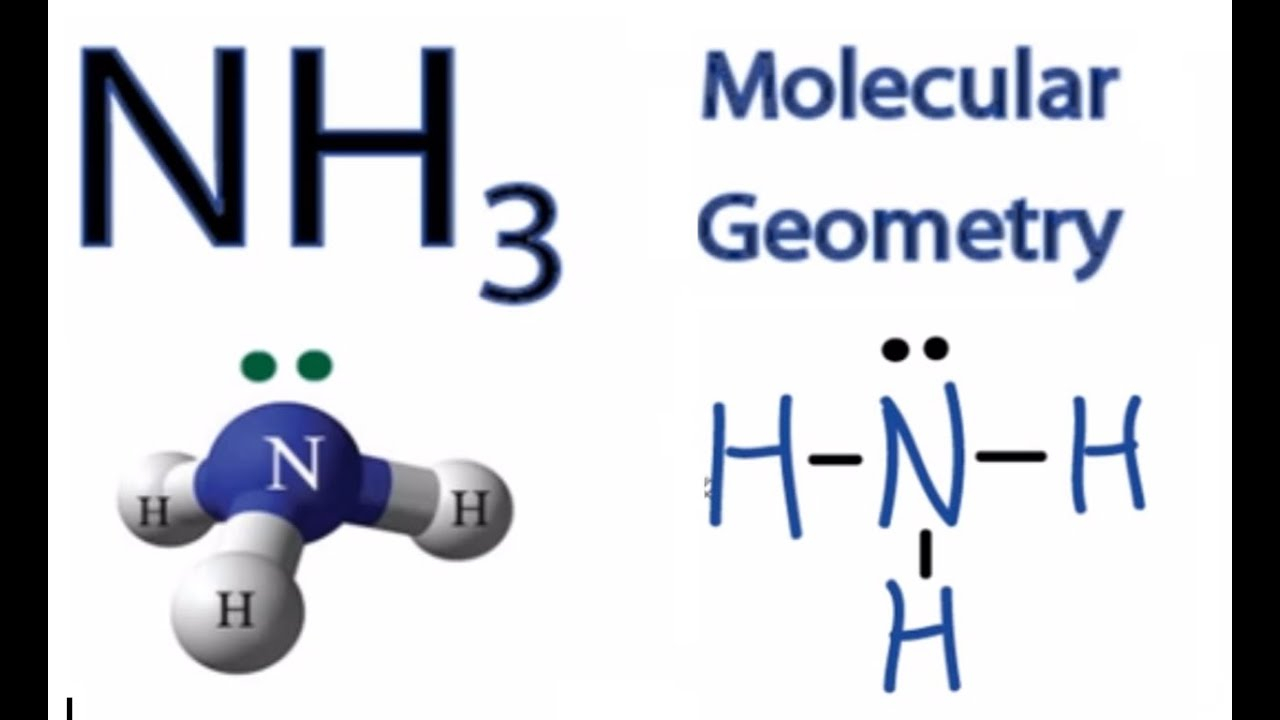
The NH3 Lewis structure is a diagram that illustrates the number of valence electrons and bond electron pairs in the NH3 molecule. The geometry of the NH3 molecule can then be predicted using the Valence Shell Electron Pair Repulsion Theory (VSEPR Theory), which states that molecules will choose the NH3 geometrical shape in which the electrons have from one another.

NH3 (ammonia) Lewis dot structure YouTube
In the NH 3 Lewis structure, there are three single bonds around the nitrogen atom, with three hydrogen atoms attached to it, and on the nitrogen atom, there is one lone pair. NH3 Lewis Structure - How to Draw the Dot Structure for NH3 (Ammonia) Watch on Contents Steps #1 Draw a rough sketch of the structure

Estructura De Lewis Del Amoníaco Nh3 Explicacion Facil Mobile Legends
The Lewis structure of nitrogen and hydrogen atom shows a total of eight valence electrons participating in a bond formation, to produce a single tetra-atomic NH3 molecule. Here, we need to study how the Lewis structure of the NH3 molecule is drawn: Search the total number of valence electrons: It is eight to form a single NH3 molecule.

electron dot structure of NH3 Science Carbon and its Compounds
A step-by-step explanation of how to draw the NH3 Lewis Dot Structure (Ammonia).For the NH3 structure use the periodic table to find the total number of vale.